- Details
Last week we exhibited at the ASCO (American Society of Clinical Oncologist) Annual meeting held in Chicago. With an estimated crowd of thirty-three thousand, the original message seemed to be dominated by immunotherapy. Everyone is looking for that trigger that will activate the immune system so the body will kill its cancer.
So why was Pyrexar Medical there? Traditionally as an adjunct to radiotherapy and chemotherapy, Hyperthermia has been very useful in shrinking tumors, killing cancer cells, and improving drug delivery.
There is preclinical evidence that heating a cancer tumor initiates a similar immuno-activation response, potentially serving as an adjunct to the immunotherapy drugs in development. The diagram below demonstrates the immunomodulation induced by hyperthermia.
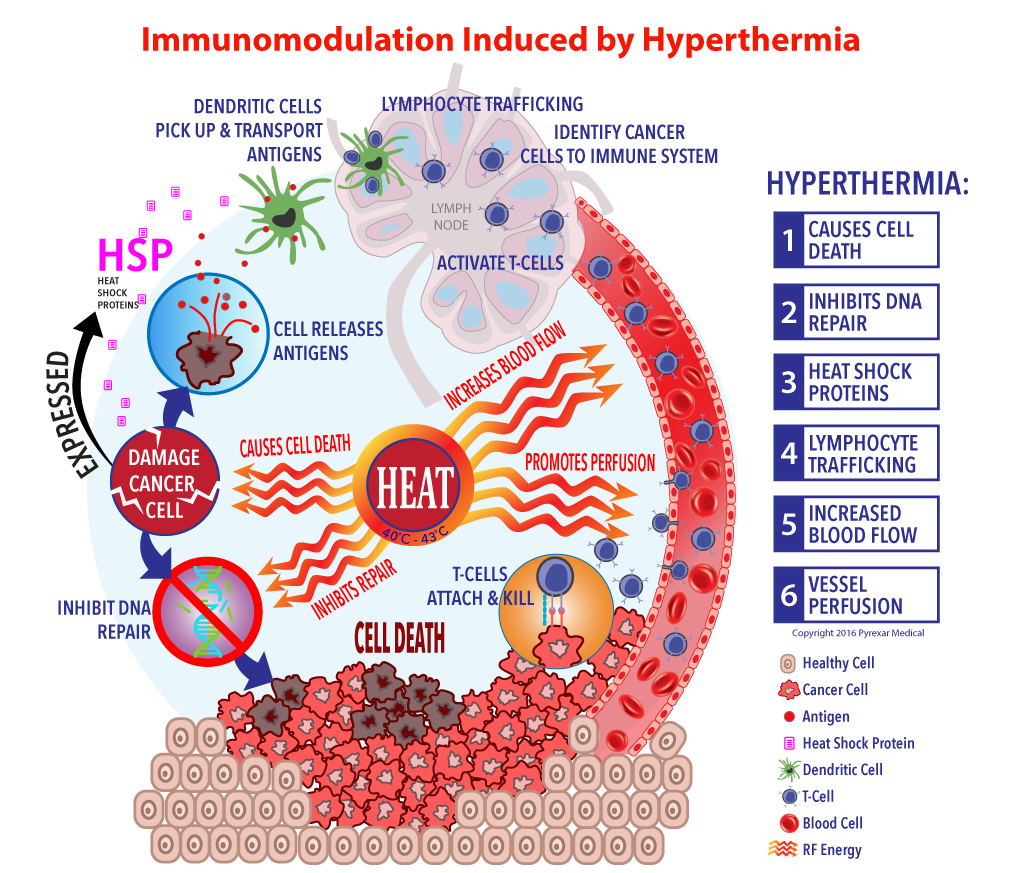
Preclinical evidence suggests that heating cancer cells cause cell stress and the release of HSP (heat shock proteins). One of the roles of HSP it to assist the delivery and integration of Antigens, molecules that induce an immune response, to the lymphatic system. With the information provided by the cancer-specific Antigen, the Lymphatic system then produces cancer-specific Tumor-infiltrating lymphocytes ( aka T-Cells) with customized receptors that are capable of identifying, targeting, and killing cancer. Once the T-cells are programmed, heating has an additional benefit of increasing blood flow, speeding up the delivery of the T-cells to the cancer site.
Immunomodulation would explain the long term survival rates as reported in a Phase III Sarcoma study “Neo-adjuvant chemotherapy alone or with regional hyperthermia for localized high-risk soft-tissue sarcoma: a randomized phase 3 multicenter study” by well-known physician Dr. Rolf Issels. In this dual armed study, half of the Sarcoma patients received chemotherapy (CT), and the other half received chemotherapy + hyperthermia (CT+HT). Sarcoma patients receiving CT+HT not only had significantly increased tumor response but remained cancer-free more than two times longer than those who received chemotherapy alone.
Heating boosts radiotherapy; heating boosts chemotherapy; it stands to reason, with all we know, that heating will boost Immunotherapy. Indeed a significant benefit for immunotherapy drug makers. Plus, this additional healing boost comes with very little to no ill effects or discomfort. Watch this short patient treatment testimonial for pancreatic cancer courtesy of ABC News Good4Utah.com





 Dr. Curt Heese, board-certified radiation oncologist at Cancer Treatment Centers of America, Philadelphia. Dr. Heese will be sharing his experiences using Pyrexar Hyperthermia systems as an adjunct therapy to treat pancreatic cancer.
Dr. Curt Heese, board-certified radiation oncologist at Cancer Treatment Centers of America, Philadelphia. Dr. Heese will be sharing his experiences using Pyrexar Hyperthermia systems as an adjunct therapy to treat pancreatic cancer. Dr. Niloy Datta, oncologist and prolific researcher in the field of hyperthermia at Kantonspital Aarau AG, Aarau, Switzerland. Dr. Niloy Data will be sharing early results from a multi-institutional phase I/II clinical study "Hyperthermia and Proton Therapy in Unresectable Soft Tissue Sarcoma (HYPROSAR).
Dr. Niloy Datta, oncologist and prolific researcher in the field of hyperthermia at Kantonspital Aarau AG, Aarau, Switzerland. Dr. Niloy Data will be sharing early results from a multi-institutional phase I/II clinical study "Hyperthermia and Proton Therapy in Unresectable Soft Tissue Sarcoma (HYPROSAR). Please welcome our new distribution partner, Clinitech Company Ltd, bringing the hyperthermia technologies into Cuba, the West Indies, Panama, and Venezuela.
Please welcome our new distribution partner, Clinitech Company Ltd, bringing the hyperthermia technologies into Cuba, the West Indies, Panama, and Venezuela.
 Just under a year ago, Pyrexar purchased the hyperthermia device group from Perseon Medical (formerly BSD Medical). As part of the original purchase agreement, Perseon acquired 19.9% of our company, along with royalties on future sales of the BSD-2000, our flagship product. Fees alone were estimated at $4 million over the next three years.
Just under a year ago, Pyrexar purchased the hyperthermia device group from Perseon Medical (formerly BSD Medical). As part of the original purchase agreement, Perseon acquired 19.9% of our company, along with royalties on future sales of the BSD-2000, our flagship product. Fees alone were estimated at $4 million over the next three years. After passing the rigorous clearance process with the TFDA (Taiwan Food and Drug Administration), a Pyrexar BSD-2000 Deep Regional Hyperthermia system is now installed and treating patients at MacKay Memorial Hospital in New Taipei City, Taiwan. On-site to provide hands-on training, and treat the first series of patients, was a well-respected physician and researcher Dr. Jacobi van der Zee from Erasmus University Hospital-Daniel den Hoed Cancer Center Rotterdam, The Netherlands. Dr. van der Zee is recognized as a world leader in hyperthermia with a long list of published papers and clinical studies.
After passing the rigorous clearance process with the TFDA (Taiwan Food and Drug Administration), a Pyrexar BSD-2000 Deep Regional Hyperthermia system is now installed and treating patients at MacKay Memorial Hospital in New Taipei City, Taiwan. On-site to provide hands-on training, and treat the first series of patients, was a well-respected physician and researcher Dr. Jacobi van der Zee from Erasmus University Hospital-Daniel den Hoed Cancer Center Rotterdam, The Netherlands. Dr. van der Zee is recognized as a world leader in hyperthermia with a long list of published papers and clinical studies.